|
It then converts the signal (like a transducer) in such a way that you get the analyte concentration. This set of electrons is then accelerated into the electron capture detector which counts electrons as it absorbs them. The free electrons travel through towards a positively-charged anode, creating a circuit. Note that the atomic number changed (1 proton) but the mass number did not change (+1 neutron). Abstract: The objective of this thesis is to expand the utility of the photodetachment-modulated electron capture detector (PDM-ECD) for the study of gas-phase. This emits radioactive electrons in the presence of, typically, #"N"_2#, which acts as a makeup gas (a gas that is used to minimize band broadening, which tends to be an issue, for instance, if you're working with many molecules with diverse root-mean-square velocities ).Īs the electron emitter emits electrons at the makeup gas, these electrons collide with them and ionize them, thus increasing the number of free electrons. Since a proton becomes a neutron, the number of protons decreases by 1, but the atomic mass stays the same. A neutral atom of Carbon-12 contains six protons, six neutrons, and six electrons therefore, it has a mass number of 12 (six protons plus six neutrons). Electron capture is a process in which a proton in the nucleus captures one of the inner electrons and forms a neutron. Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. Typically, about #10#"mCi"# (a measure of radioactivity, called "Curies" after Marie Curie #"mCi"# is a millicurie) of the #""^63 "Ni"# radioactive nuclide acts as an electron emitter. Rubidium-83 will decay into krypton-83 by electron capture. 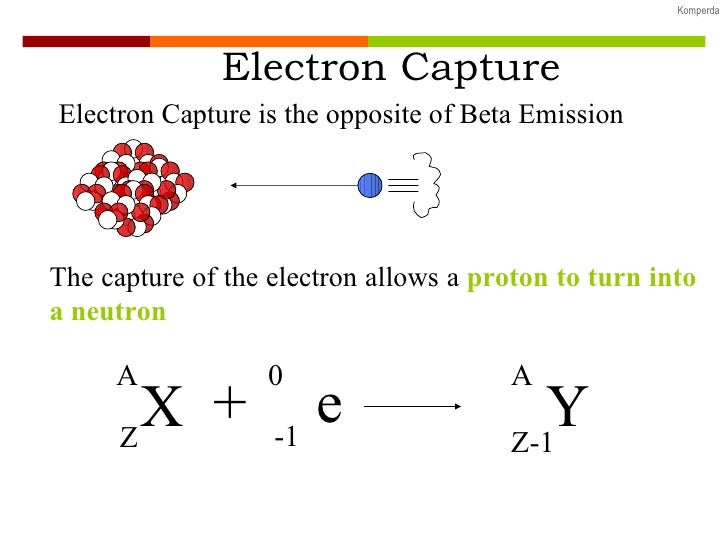 
Suppose we had a gas chromatograph that sent its output stream of carrier gas (the mobile phase) through the column labeled #A# below: We consider capture cross sections from hydrogen to the nl-subshell of bare-ion projectiles calculated within the EI approximation. In general, the purpose of an electron capture detector is to detect the concentration of the analyte based on how many electrons it absorbs to use in an electron capture event. Miraglia, Maria Silvia Gravielle, in Advances in Quantum Chemistry, 2022 3.2 Electron capture cross-section data set.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
